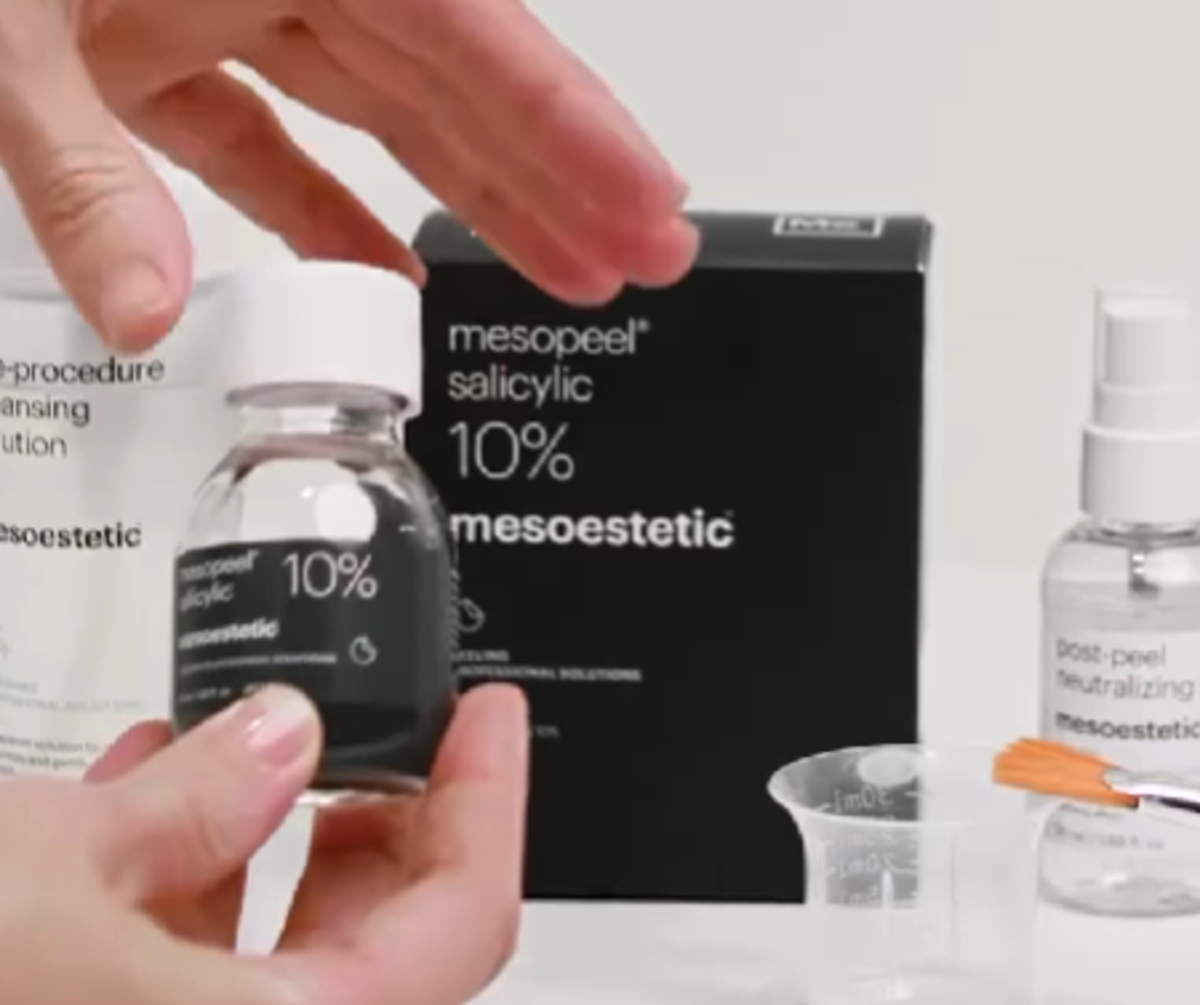
Salicylic acid product by Mesoestetic. Credit: Instagram @Mesoestetic
Spain’s luxury skincare brand Mesoestetic is facing regulatory action in India after several of its products were found to contain salicylic acid at unsafe levels, far exceeding limits set by Indian cosmetic laws.
The Central Drugs Standard Control Organisation (CDSCO) officially cancelled the registration of multiple Mesoestetic products on May 20, 2025, due to non-compliance with Indian safety standards.
Mesoestetic: Salicylic acid levels up to 30% found
Under India’s Cosmetics Rules, 2020, salicylic acid is permitted at:
- 2 per cent for general cosmetics
- 3 per cent for rinse-off hair products
But the CDSCO found some Mesoestetic products contained between 7 and 30 per cent, prompting serious health concerns. The affected items include:
- Mesopeel Periocula – 7 per cent
- Mesoestetic Salicylic (10 per cent, 20 per cent, 30 per cent)
- Dermamelan Intimate Gel – 15 per cent
- Dermamelan Mask – 15 per cent
- Mesopeel Modified Jessner – 14 per cent
These were imported by Spectra Medical India Pvt. Ltd, based in Tamil Nadu, Mesoestetic’s official Indian partner.
The CDSCO issued a show-cause notice on April 7, 2025, after lab tests confirmed the violations. Spectra Medical India responded by acknowledging the breach and agreeing to surrender the products.
The dark side of salicylic acid: Salicylate toxicity
Health experts warn that excessive exposure to salicylic acid – especially in high concentrations – can lead to salicylate toxicity, a potentially life-threatening condition.
Early symptoms include:
- Nausea and vomiting
- Ringing in the ears (tinnitus)
- Headaches and dizziness
- Abdominal pain and excessive sweating
- Rapid or deep breathing
Severe cases may result in:
- Confusion or hallucinations
- Seizures or coma
- Kidney failure and respiratory distress
- Brain swelling and abnormal heart rhythms
Prompt medical attention is crucial if symptoms appear after using products containing high salicylic acid levels.
India’s booming skincare market
India’s luxury skincare sector is booming, valued at $8.78 billion (€8.08 billion) in 2024 and projected to reach $17.69 billion (€16.28 billion) by 2033, with e-commerce platforms like Nykaa and Tira fuelling demand for premium international brands. (As reported by South First.)
However, the CDSCO crackdown signals tighter scrutiny. According to Dr Rajeev Singh Raghuvanshi, India’s Drugs Controller General, the cancelled Mesoestetic products violated Rule 39 of the Cosmetics Rules, 2020, and Condition 03 of their registration certificate. (As reported by pharmabiz.com.)
Mesoestetic skincare, Spain
Mesoestetic, headquartered in Spain, has a presence in over 105 countries and reports that 87 per cent of its sales come from international markets, according to its official website.
Based on the SCCS Final Opinion on Salicylic Acid (CAS No. 69-72-7), which applies across all EU member states, including Spain, the legal concentration limits for salicylic acid in cosmetic products under Regulation (EC) No. 1223/2009 are as follows:
- Up to 3.0 per cent: Allowed in rinse-off hair products.
- Up to 2.0 per cent: Permitted in other cosmetic products, except the following:
- Body lotion
- Eye shadow
- Mascara
- Eyeliner
- Lipstick
- Roll-on deodorant
- Up to 0.5 per cent: Allowed in the above six exceptions, as well as in:
- Oral products (only for inhibiting microbial development, which must be clear from the product presentation)
- Non-spray deodorants
As luxury skincare demand grows across India and beyond, regulators are making it clear that even premium brands aren’t exempt from violating safety guidelines.
View all health and beauty articles.
Check out alternative and natural skincare.







